As you age you will see a reduction in Testosterone levels – which is normal. What’s not normal is feeling like an absolute ass because of it.
Look, I get it. You don’t wanna go on TRT but you definitely want better hormone levels in your own body. God forbid you wanna feel like you should feel.
Luckily, there are ways we can improve hormones, so let’s see How To Naturally Increase Testosterone.
Key Takeaways
- Low testosterone may affect the quality of life.
- Testosterone replacement therapy is not recommended if you still want to have children.
- Talk with your doctor about your best options and ask about how to increase testosterone (T) levels naturally.
- Always keep in mind that better lifestyle always leads to a better quality of life.
- Sleep well, eat well and work out… This is your way to go.
Testosterone Deficiency
Serum testosterone levels decline gradually with age at an approximate rate of 1% per year after the third decade of life.
The American Urological Association (AUA) defines hypogonadism, or testosterone deficiency as a total testosterone level less than 300 ng/dL on early morning blood draw on two separate occasions. [1]
Testosterone deficiency is highly prevalent, with rates increasing with age from 4% in men aged 40–49 years to nearly 50% in men over 80 years of age, with some studies suggesting a 17% increase in low testosterone levels for every decade of life. [1]
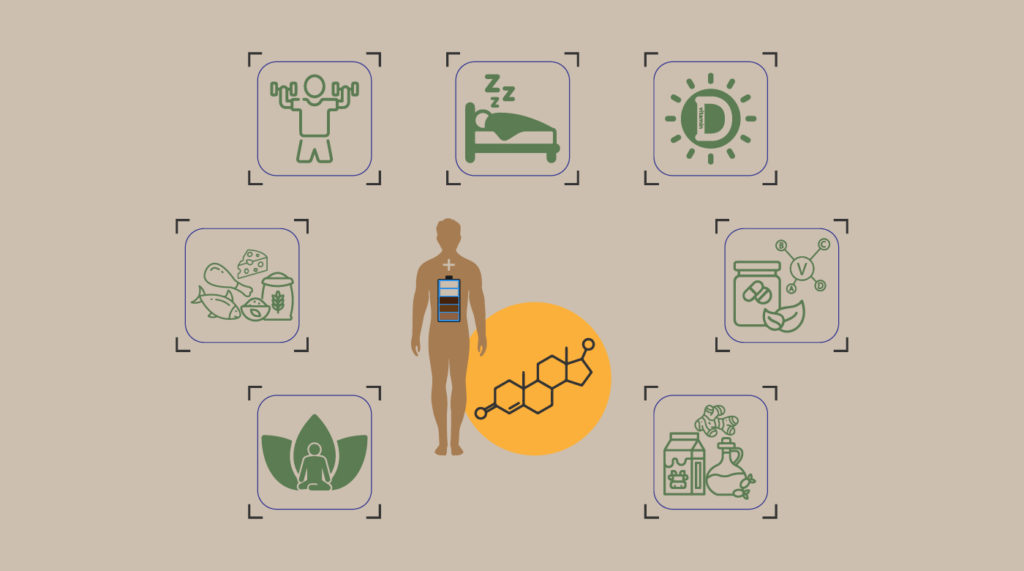
How To Naturally Increase Testosterone
Lifestyle modifications
In general, lifestyle modifications to include healthy sleep with healthy diet and exercise to maintain a normal weight and activity level are safe and effectively raise endogenous testosterone levels. These recommendations should be included as part of the counseling and management options for men with testosterone deficiency. [1]
While it may not always be possible to live a completely stress-free life, you can do some things to lower stress levels and therefore boost testosterone levels.
Maintaing Healthy Weight and Lipid Profile
Low levels of testosterone can negatively affect body composition, and as people age, reduced testosterone levels can also cause weight gain.
Studies have shown that both low testosterone and obesity increase the risk of major adverse cardiac events. [1]
Recent preliminary data have shown that low endogenous testosterone levels are associated with lower high-density lipoprotein (HDL) cholesterol and higher low-density lipoprotein cholesterol, triglyceride, and total cholesterol levels. [1]
As such, the AUA recommends counseling men with testosterone deficiency to increase physical activity and maintain their weight within the recommended range to reduce the signs and symptoms of testosterone deficiency and increase serum testosterone levels. [1]
Armamento- Villareal and colleagues reported that dietary interventions, with and without exercise, resulted in significant increases in total testosterone levels. [11]
Similarly, another study reported on 68 men participating in a weight-loss diet. Men lost a mean of 10.3–10.8±1.2 kg over the 52-week study period and experienced significant increases in total and free testosterone (P<0.001 and P=0.002, respectively). [1]
Additionally, men with moderate to severe baseline erectile dysfunction had significant increases in the International Index of Erectile Function (IIEF) erectile function domain following weight loss. [1]
More recently, Rigon and colleagues evaluated 29 men with a mean baseline weight of 155.26±25.88 kg preoperatively and 6 months postoperatively following bariatric surgery. [1]
BMI improved to a mean of 37.82±5.94 kg following surgery, with improvements in total testosterone levels from 229.53±96.45 ng/dL to 388.38±160.91 ng/dL (P<0.001). [1]
Vitamin D

Vitamin D is an important nutrient naturally found in food such as mushrooms and fatty fish (i.e. sardines, salmon, mackerel) in addition to milk (fortified with vitamin D).
The body does produce high amounts of vitamin D when skin is directly exposed to sun (sunshine vitamin). However, excess sun exposure may increase the risk of skin cancer.
Because vitamin D is difficult to find in foods and prolonged sun exposure carries health risks, dietitians and doctors often prescribe supplements of varying doses.
The male reproductive tract has been identified as a target tissue for vitamin D, and previous data suggest an association of 25-hydroxyvitamin D [25(OH)D] with testosterone levels in men.
It’s a natural testosterone booster because it plays a crucial role in hormone production.
Exercise
A previous study showed that serum testosterone, glycated hemoglobin (A1C), fasting glucose, high‑density lipoproteins (HDL), triglycerides (TG), and waist circumference were significantly improved after 52 weeks of supervised diet and exercise without testosterone treatment. [13]
There is epidemiologic evidence that frequent vigorous exercise is associated with a 30% lower risk of erectile dysfunction. [13]

Moderate workout regimen reduces the risk of ED by two‑thirds, and in men with high physical activity, erectile dysfunction. is reduced by over 80%. [13]
The serum testosterone levels of obese men have been reported to be significantly increasing following a 12‑week course of regular aerobic exercise and dietary regulation. They explained that the aerobic exercise and dietary regulation reduced the insulin levels of obese males, leading to increased serum testosterone levels. [13]
Sleep Quality
Aside from maintaining a healthy weight, sleep quality has been associated with symptoms of testosterone deficiency. [1]
Pastuszak et al. noted a linear relationship between self-reported sleep quality and symptoms of testosterone deficiency as quantified by the Androgen Deficiency in the Aging Male (ADAM) questionnaire. [12]
Most studies suggest a correlation between sleep quality and symptoms of testosterone deficiency. [1]
Herbal supplements
Many herbal supplements are also marketed towards “boosting” testosterone levels, including onion (Allium cepa L.), ginger (Zingiber officinale), maca root (Lepidium), fenugreek (Trigonella foenum-graecum), and black seeds (Nigella sativa). [1]
Onion
Onion is thought to work by increasing the production of LH and reducing testicular reactive oxygen species (ROS). [1]
Ginger
Ginger has been hypothesized to improve testosterone release by reducing ROS in the testis. [1]
Fenugreek Seeds
Fenugreek seeds have been touted to have a wide range of health benefits and anti-inflammatory properties and have been used to treat women’s health conditions, diabetes, and hypercholesterolism. [1]
It has been suggested that this compound also improves testosterone levels and symptoms associated with testosterone deficiency. [1]
This compound is thought to have androgenic effects via its soluble steroidal saponin content and incomplete 5-alpha reductase and aromatase inhibitor (AIs) activity. [1]
A study by Maheshwari et al. treated 50 men aged 35–65 diagnosed with symptomatic hypogonadism with 500 mg of daily fenugreek seed extract for 12 weeks. Subjects experienced a significant increase in free testosterone (8 vs. 12 pg/mL, P=0.004), but no significant increase was noted in total testosterone (405 vs. 436 ng/dL, P=0.16), following 12 weeks of treatment. [9]
Improvements in sperm count and motility were also noted after 4, 8, and 12 weeks of treatment, with improvements in sperm morphology, noted at 8 and 12 weeks. [9]
No changes were noted in DHEA levels, fasting blood sugars, liver enzymes, cholesterol levels, or total leukocyte counts. [9]
This treatment appears to be well tolerated and effective, but long-term data regarding safety and efficacy are lacking, and further studies need to be performed before the establishment of fenugreek as a viable option for treating testosterone deficiency. [9]
Zinc
Zinc deficiency has been linked to decreased spermatogenesis and testosterone production. [1]
Liu et al. randomized 67 men with isolated hypogonadotropic hypogonadism to receive FSH or FSH plus 40 mg zinc for 18 months. [10]
They found no differences in serum testosterone levels between groups throughout the study period. Similarly, studies suggest that the use of zinc alone is ineffective in increasing testosterone levels. [1]
Overall, while certain herbal supplements appear to be well tolerated, with preliminary data suggesting increases in testosterone levels, prospective randomized controlled trials in humans must be conducted to demonstrate safety and efficacy before these compounds can be supported for use in the
Who Is Eligible For Testosterone Replacement Therapy (TRT)?
The AUA Panel on the Evaluation and Management of low testosterone suggests that to be eligible for testosterone therapy patients must have:
- Total testosterone level less than 300 ng/dL on early morning blood draw on two separate occasions. [1]
- Symptoms of low testosterone such as anemia, mood swings, Erectile dysfunction, Decreased libido and sexual function, reduced muscle mass and strength, reduced bone mass and osteoporosis, insomnia and a reduced sense of general well-being, forgetfulness, difficulty in concentration, and loss of memory. [1]
Testosterone Deficiency and TRT
Testosterone deficiency has been treated with various formulations of exogenous testosterone. Exogenous testosterone, however, bypasses the body’s natural ability to produce testosterone, thus leading to the downregulation of luteinizing hormone (LH) and follicle stimulating hormone (FSH) with suppression of the hypothalamic-pituitary-gonadal (HPG) axis and spermatogenesis. [1]
Side Effects of Testosterone Replacement Therapy
Injectable testosterone can result in fluctuations in serum testosterone levels. [1]
Alternative formulations of exogenous testosterone, such as gels or creams, have the risk of transference to others. [1]
Testosterone therapy may also lead to polycythemia. [1]
Erythrocytosis is a common adverse effect of TRT, particularly with testosterone ester injections and in older men. Middle-aged and older men on TRT are nearly four times as likely to have HCT > 50%.
TRT increases prostate volume and PSA. There is unequivocal evidence that TRT can stimulate growth and aggravate symptoms in men with locally advanced or metastatic prostate cancer.
The Endocrine Society and American Urological Association (AUA) recommend against the use of testosterone for treatment of hypogonadism in men who desire fertility in the next 6 to 12 months.
Previous studies suggested that there is an increased risk of venothromboembolic (VTE) events in the first six months of treatment, and warnings regarding cardiovascular and embolic events remain in the drug packaging information. [1]
Given the side effects and controversies of exogenous testosterone therapy, much work has been done to identify effective alternatives to treat symptomatic testosterone deficiency. [1]
Here are some alternatives to exogenous testosterone that can boost testosterone naturally. [1]
management of testosterone deficiency. [1]
TRT Treatments
Selective Estrogen Receptor Modulators (SERMs)
SERMs block the negative feedback of E2 on the hypothalamic pituitary axis, resulting in increased LH production and increased downstream testosterone production. [1]
These medications are orally administered which improves patient compliance. [1]
Clomiphene Citrate
Clomiphene citrate has been used as a treatment to restore testosterone levels and promote or preserve spermatogenesis in men with testosterone deficiency desiring to preserve fertility. [1]
This medication is an oral compound administered at doses ranging from 25 mg every other day, to 50 mg daily, based on testosterone response. [1]
Moskovic et al. reported on 29 men with a baseline total testosterone level of 228±48 ng/dL treated for >3 years with clomiphene citrate. [2]
They demonstrated improvements in testosterone to a mean of 582±227 ng/dL (P<0.001), as well as significant improvements in ADAM questionnaire response (P=0.01) and mean BMI (P<0.05). [2]
Similarly, Taylor and Levine demonstrated that compared to testosterone gel therapy, clomiphene citrate treatment resulted in equivalent, significant improvements in testosterone levels at a fraction of the cost, as well as significant improvements in ADAM questionnaire scores. [3]
Additionally, patients on clomiphene citrate treatment did not experience changes in hemoglobin, PSA, or cholesterol levels while on treatment. [3]
Ramasamy et al. have also demonstrated that quantitative ADAM scores were similar among patients treated with clomiphene citrate as compared to those treated with testosterone gels or injections, with improvements in testosterone levels while on clomiphene citrate treatment to physiologic levels. [4]
Rare side effects include headaches, visual changes, and gynecomastia (abnormal breast enlargement). [1]
Additionally, clomiphene citrate use has shown to have significantly less risk of increased hematocrit as compared to testosterone therapy. [1]
Preliminary data on men treated for testosterone deficiency on clomiphene citrate for as long as 7 years show that over 80% of men achieve testosterone levels >450 ng/dL, with 78% of men reporting subjective improvement in hypogonadal symptoms, and only 9% reporting side effects from clomiphene citrate treatment with no significant adverse events. [1]
This data together suggests that clomiphene citrate may be a safe and effective treatment option for the long-term management of testosterone deficiency, and consideration should be made to offering this therapy to men as a first-line treatment option for testosterone deficiency, regardless of the patient’s desire for fertility preservation. [1]
Tamoxifen
Like clomiphene citrate, tamoxifen has been shown to increase testosterone and gonadotropin levels, and preserve spermatogenesis. [1]
This SERM is an acknowledged alternative treatment option for testosterone deficiency in men. [1]
Tamoxifen has been used to stimulate gonadotropin production, and to treat gynecomastia in the setting of anabolic steroid-induced hypogonadism or hCG treatment. [1]
However, the adverse effects associated with tamoxifen appear to be greater than those associated with clomiphene citrate use, including gastrointestinal distress, venous thromboembolic events, and other cardiovascular outcomes. [1]
As such, tamoxifen is less commonly used for the treatment of testosterone deficiency. [1]
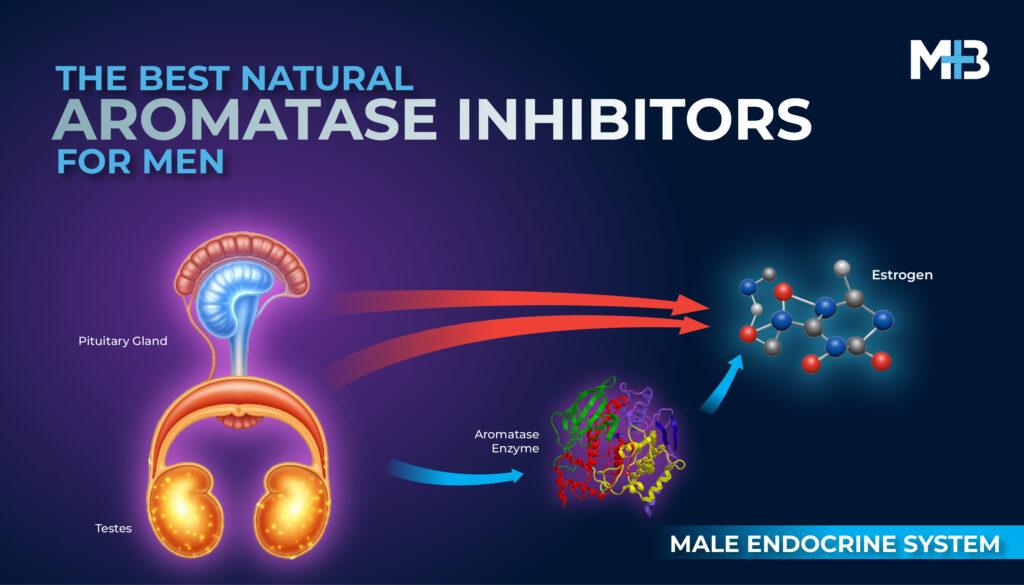
Aromatase Inhibitors
You’re probably familiar with the term “testosterone.” While the female hormone estrogen is also present in males.
Like SERMs, aromatase inhibitors (AIs) such as letrozole and anastrozole, are also designed to block E2 inhibition of the HPG axis by preventing aromatization of testosterone to E2 in the tissues, thus upregulating endogenous testosterone production. [1]
These compounds have been shown to raise testosterone levels in hypogonadal men and are also orally administered. [1]
Dias et al. treated 14 hypogonadal men with 1 mg of anastrozole daily for 12 months. Men treated with AI had significant increases in total testosterone levels over the study course.
After 12 months, fasting glucose, insulin, and lipid levels were not different between men treated with AI and men treated with testosterone therapy. [5]
AIs are well-tolerated, but side effects may include nausea, the elevation of liver enzymes, headaches, and hot flashes. [1]
As long-term AI use has been associated with decreases in bone mineral density due to suppression of E2, the AUA recommends against the use of AI for prolonged periods. [6]
hCG
hCG is an FDA approved LH analog that stimulates testosterone production by binding LH receptors. [1]
hCG has also been used with concurrent exogenous testosterone to maintain intratesticular testosterone levels and preserve spermatogenesis. [1]
Liu et al. randomized 40 hypogonadal men over the age of 60 to receive either 5,000 IU of hCG or placebo twice per week for 3 months. Patients treated with hCG had significant increases in total and free testosterone. [7]
More recently, Habous et al. treated 94 men with 5000 IU of hCG twice per week for 3 months. They similarly found significant increases in testosterone levels with hCG treatment. [8]
While hCG acts directly on the testis and is well-tolerated, this medication is expensive, and with the requirement for multiple injections per week, many patients elect alternative treatments. [1]
Skip Ahead
Toggle