Certificates of Analysis. What does it actually mean, and how do you even know how a good one looks?
We see a COA in every single SARM and Peptide supplier, and we refer to these “tests” to justify us using said supplier – but could you even identify a fake one?
Doing testing is essential, especially to find consistent services from a supplier. The COA is a reflection of that testing and their methodologies for creating a said product.
Problem is, COAs are incredibly hard to understand – but they don’t have to be.
Key Takeaways
- A certificate of analysis allows the seller to show the client that their products are of high quality
- A COA is preferable for all sales of SARMs, Peptides, and other Research Chemicals
- A COA is done with the HPLC (High-performance liquid chromatography) method with UV and electrospray ionization – mass spectrometry (ESI–MS)
What are COAs?
A COAs or Certificate of Analysis is a piece of documentation that will show the end consumer a bunch of information about a certain product. This is one of the resources they can use to be transparent about who they are, how good their production process is, and how pure their product is as well. A COA should usually include the following:
- Supplier Information: Pretty self-explanatory, but this is just a collection of information regarding the producer of the SARM or Peptide
- Tester Information: Since testing is to be done by a third party, there should be a good amount of information to cover who the tester is. It is also advised to do research on the tester to make sure they are legit
- SARM/Peptide Identification: They have to clarify which SARM or Peptide they will be testing so that you may reflect on the data
- Test Results: Arguably the most important piece of information on any of these documents. This is what you pay a higher price for, and good management would have the supplier test batches regularly
- Signature: At the end of the documentation there should be some signature or electronic signature to ensure the tests were done truthfully
Now that you have a general idea of what should be on one of these tests, you should be able to manage to see a good one from a bad one. That said, in the developing world of SARMs and Peptides there are bound to be companies that try to get by cheaply. But what do COAs actually test for? They test for the purity of a product via HPLC.
What is COA Testing?
For most COA testing (for SARMs and Peptides) a detection called the HPLC (High-performance liquid chromatography) method with UV and electrospray ionization – mass spectrometry (ESI–MS) will be used.
A simple summary of HPLC is “High-performance liquid chromatography (HPLC) is a technique used to separate molecules based on size and surface charge, among other properties. “
Mass spectrometry, the “last part”, will identify the molecular weight of a certain compound. For instance, Hydrogen (H) has a molecular weight of 1,00784 u, whereas Oxygen (O) has a molecular weight of 15,999 u. Thus, mass spectrometers can be used to identify unknown compounds via molecular weight determination. All mass spectrometers will use three different practices to do this, namely:
- Ionization Source
- Mass Analyzer
- Ion Detection System
If you know the weight of certain molecules, you can calculate their concentration of them with simple mathematics and they will also use the Beer-Lambert Law and UV light to calculate this.
How to read COAs?
Now that you (hopefully) have an understanding of how the HPLC method works, we can start looking at how you read a COA. You’ve seen all the graphs with one huge spike and that “supposably” means the product is pure. Cool!
But how does that even work?
The left verticle line (the Y-axis) will read mUa (milli absorbance unit). The lower horizontal line (the X-axis) will read t (time).
mUa represents the concentration by showing the absorption of analytes. Time is simply the time of retention (time from “inserting” the compound to the detection of the compound).
They will then use a reference list to review and see which compound is found. For instance, Mk-677 might have a retention time of 6 minutes and 45 seconds. Thus, if the compound is detected at 6 minutes and 44.98 seconds, it’s most likely Mk-677.
Lastly, the higher the spike on the graph, the higher the concentration of the said compound. For more info, the video above gives a very detailed description.
Thus, you should be looking for a COA of a compound that has a retention time that aligns with the reference, and also has a very high concentration.
How to see if a COAs is Fake?
There is a great risk that a COA might be faked for the purpose of getting you to purchase the product. If they can use the “support” of a “good” COA, any company would make this decision. Since research chemicals are not governed by any organizations, who’s to stop them? Here are some of the things you need to look out for:
- Dates that are way too old. This is something companies do to cut down costs, and it simply means they do not test every batch. It is the problem we see most often when it comes to SARMs and Peptides, and if they’re unable to even test the purity, how could you trust them to make pure stuff?
- See if the retention time matches others. Since the retention time is similar for all of the same compound, finding one that does not fit the others is the fastest way to see if a product is bogus.
- If the graph has too many spikes it could be an indication that there are too many contaminants in the compound. An example of this would be cheap packaging or preservation chemicals.
- Who did the testing. I have done a bit more research on one or two “companies” managing the testing and turns out they don’t exist. The retailer simply made the results in Photoshop. So, do some research…
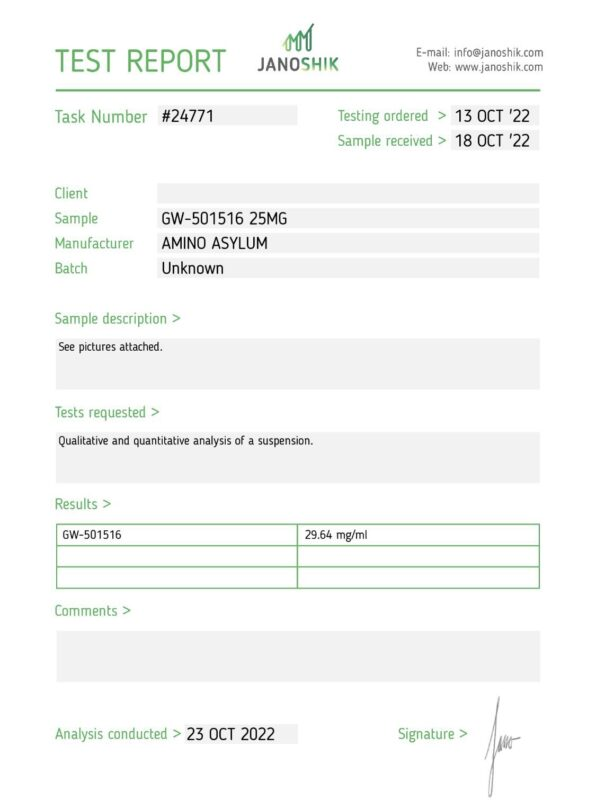
Let’s have a look at the figure above. First off, the test is several months old. Secondly, when you Google JanoShik, you’re actually met with a real site. However, the company puts a unique key at the bottom of each test to allow people to verify their tests.
This key, as you can see, is not available above. The batch number is missing, and the results merely indicate that there is GW-501516 in the sample… but nothing else, not purity, nothing.
Is this a reliable test report? Anything but.
Which companies provide COAs?
- Sports Technology Labs
- Biotech Peptides (upon request)
- Swiss Chems
Conclusion: COAs – A Practical Guide
COAs (Certificates of Analysis) help you make informed decisions about which companies deserve your support.
If you’re considering research chemicals, choose companies that invest in high-quality testing. Their commitment to quality ensures you get safe and effective products.
Research the compound, retention time, and testing company to verify their credibility. Understanding these details helps you spot reputable suppliers and avoid unreliable ones.
Taking the time to dig deeper can uncover valuable insights that benefit you for years. A little research now could save you headaches later.
FAQs about COAs
What is the use of a certificate of analysis?
The use of a COA is to let the end user know the quality of the product being purchased. When it comes to products being consumed “estimates” are no good, and being able to grant your clientele the assurance that you sell pure products will pay back tenfold.
How much does a Certificate of Analysis cost?
It depends on where you have the test done and on what the test is done.
Skip Ahead
Toggle